HOW CAN WE HELP?
Call us at 860.747.6333 or submit an information request form and someone will get back to you within 48 hours.
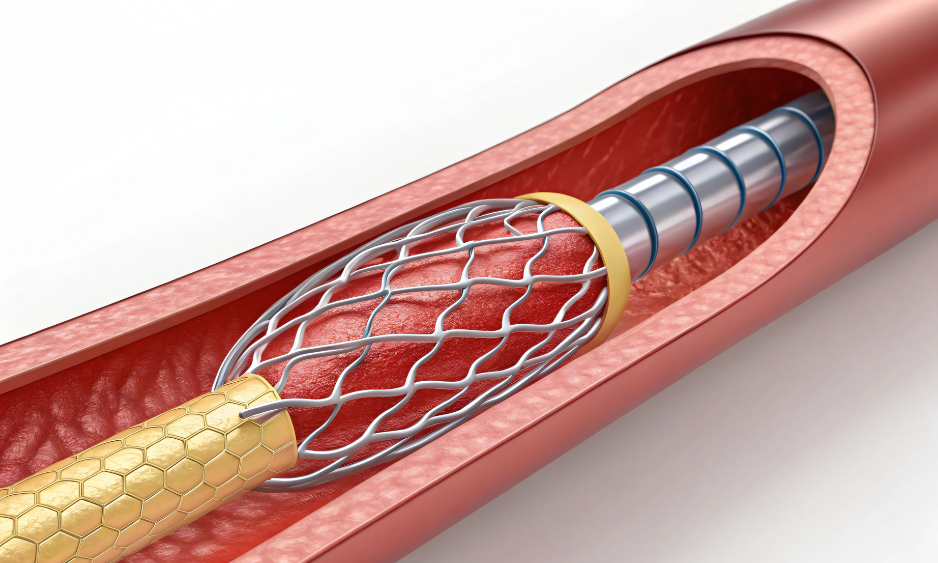
If your device is struggling with inconsistent flow, pressure spikes, or miniaturization constraints, you’re not alone. In laparoscopy, implantable pumps, surgical robotics, and IVD systems, inconsistent flow and inadequate filtration are the root causes behind performance drops, failed validations, and long time-to-market.
How Mott + IDEX help:
We engineer mission‑critical components – from porous metal/ceramic filtration and restrictors to microfluidic manifolds and hermetic assemblies – that deliver stable, laminar flow, clog‑resistance, and clinical‑grade durability. Then we back it up with rapid prototyping, in‑house testing, and a prototype‑to‑production pathway built for speed.
Consider Mott’s controlled release drug diffusion testing. We arrange and conduct drug diffusion studies to guide design criteria for your implantable controlled release technology with our proven membrane.
ISO/AS9100/ITAR,
99% on‑time delivery,
global tier‑1 OEM track record
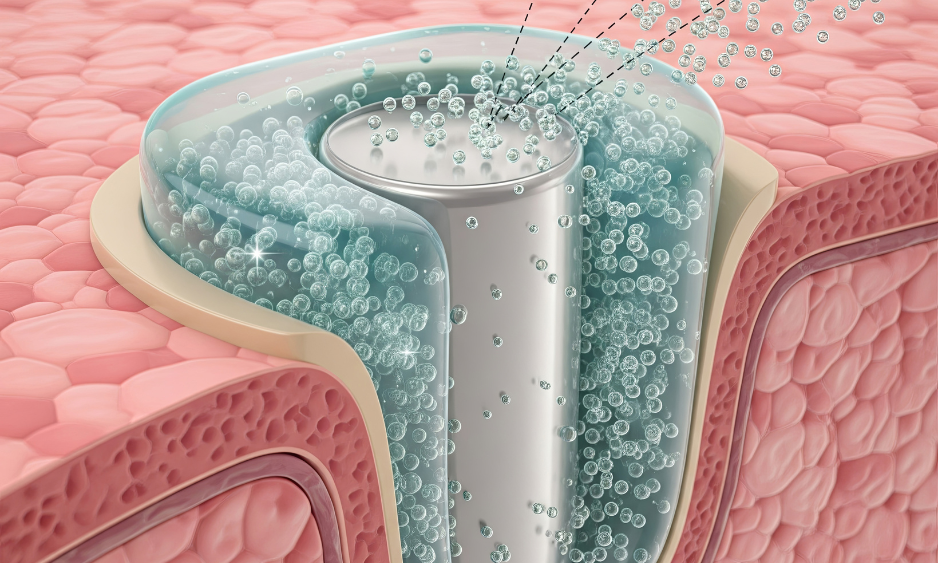
 Clog resistance & contamination prevention
Clog resistance & contamination prevention
 Designed for harsh & extreme conditions
Designed for harsh & extreme conditionsApplication specific consulting (porosity, geometry, flow rates, cost optimization.
Prototypes in days, iterative lab testing for flow, pressure, filtration, and thermal stability.
Performance data, material compatibility, and clog resistance verification.
High volume manufacturing with clinical grade quality and on time delivery.
Continuous improvement to extend product life and reduce per procedure cost.
Mott’s MedTech brochure highlights precision porous metal solutions engineered for medical devices and life‑science applications. Learn how controlled porosity, material integrity, and custom design support drug delivery, diagnostics, and advanced therapeutic systems where reliability and consistency are critical.
This case brief examines how engineered porous metal components improve flow stability in cryoablation systems. See how Mott’s solution mitigates ice formation, maintains consistent flow, and supports reliable thermal performance in critical medical procedures.
Mott’s in‑vitro drug diffusion testing supports the early design of implantable drug delivery devices. Learn how controlled release studies help estimate therapeutic windows, optimize drug concentrations, and guide volumetric and material design decisions using proven porous diffusion technology.
Mott’s in‑vitro drug diffusion testing supports the early design of implantable drug delivery devices. Learn how controlled release studies help estimate therapeutic windows, optimize drug concentrations, and guide volumetric and material design decisions using proven porous diffusion technology.
This case study demonstrates how Mott’s porous diffusion technology enables long‑term, controlled delivery of monoclonal antibody therapies through implantable drug delivery systems. Learn how precise diffusion control improves dosing consistency and supports patient‑centric treatment approaches.
Our solution: Cryo‑stable porous filtration + optimized flow restrictors for constant thermal delivery, anti‑clog performance, and gamma‑sterilizable designs.

Our solution: Tight‑tolerance orifice plates, precision microchannels, degassing systems, and biocompatible materials to ensure consistent, ultra‑low flow rates.

Our solution: Biocompatible porous filtration and metered flow restrictors that maintain predictable delivery and resist clogging and increase durability throughout device life.

Our solution: Micro‑components, hermetic ceramic‑to‑metal feedthroughs, and filtration assemblies that fit tight geometries that ensures the highest quality and performance of medical devices.

Our solution: Optical filters & illumination integrated with microfluidics for clean, precise flows and reliable visualization.



PROVEN CLINICAL PERFORMANCE
VALIDATED. RELIABLE. LONG‑TERM.
STABLE FLOW IN CRITICAL PROCEDURES
Q1. We see clogging in cryoablation laparoscopic procedures during longer procedures. Can filtration alone fix it?
Q2. Our IVD microchannels are inconsistent at ultra low flow rates.
Q3. How do you handle sterilization and biocompatibility?
Q4. Can you scale from prototypes to production?
Seamless transition from early design and rapid prototyping to scaled production, helping MedTech teams reduce risk, shorten timelines, and accelerate time to market
Proven performance in mission‑critical applications where consistency, reliability, and repeatability directly impact outcomes – from spaceflight to medical devices.